2023 Judith H. Greenberg Early Career Investigator Lecture
Lard of the Flies: Investigating How Fat Cells Communicate With the Brain
 Credit: Robert Hood at Fred Hutch.
Credit: Robert Hood at Fred Hutch.Akhila Rajan, Ph.D.
Associate Professor
Basic Sciences Division
Fred Hutchinson Cancer Center
Lecture followed by Q&A session
National Institutes of Health
Bethesda, Maryland
*NIH students and trainees working at the main NIH
campus are encouraged to attend in-person
Research Summary
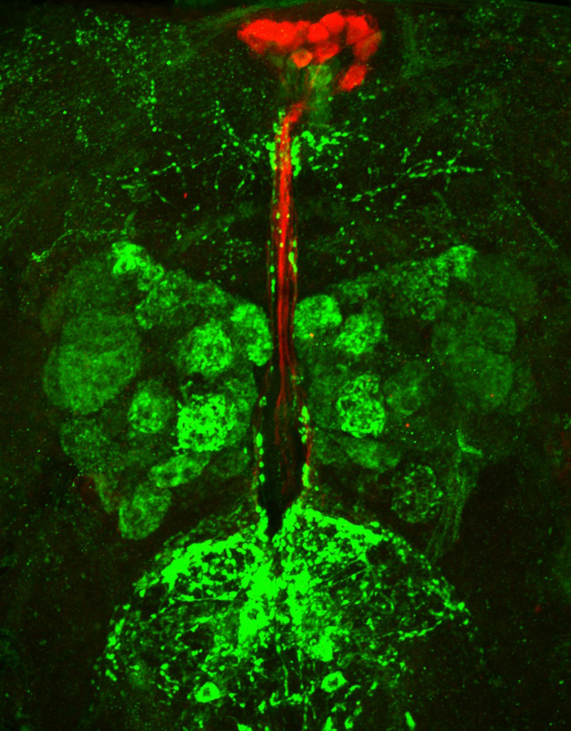 A confocal microscopy image showing a Drosophila adult brain. Fat sensing neurons are in green, and insulin-producing neurons are in red. The fat talks to the brain via adipokine signaling to the green neurons. Credit: Dr. Akhila Rajan.
A confocal microscopy image showing a Drosophila adult brain. Fat sensing neurons are in green, and insulin-producing neurons are in red. The fat talks to the brain via adipokine signaling to the green neurons. Credit: Dr. Akhila Rajan.The brain relies on input from the body to make effective decisions, such as whether to expend energy on foraging for food or use it to increase immunity and reproductive fitness. Fat cells, or adipocytes, release signaling molecules called adipokines that function as an “adipometer” and alert the brain that energy is available. A drop in levels of the fat-signaling adipokine called leptin signals starvation, allowing the brain to adapt organismal behaviors to survive adverse nutrition states.
During Dr. Akhila Rajan’s postdoctoral work, she studied the fruit fly (Drosophila) as a research organism. She identified a signaling molecule in the fruit fly that is structurally similar to human leptin, disproving the belief that leptin evolved in vertebrates. This deep and unexpected evolutionary conservation of fat-brain adipokine signaling between humans and fruit flies down to the molecular and neural circuit level suggests that Drosophila can provide significant insights into the molecular underpinnings of energy sensing and communication.
In her research lab, Dr. Rajan uses the fruit fly to understand how adipocytes control brain function, with a particular emphasis on questions that have proven to be more difficult to answer in mammalian models. Through an interdisciplinary approach, including systems-level proteomics and lipidomic analyses, powerful genetics, and whole-organism energy physiology studies, her lab’s work has provided unexpected insights into cell and neurobiological mechanisms regulating energy balance. Her lab has uncovered how fat-sensing neural circuits maintain body weight and how high-sugar diets disrupt feeding behavior and response to injury in nerve cells. Going forward, she is investigating nonhormonal mechanisms of fat-brain communication, such as lipids, organelle components, and antimicrobial peptides.
Biographical Sketch
Dr. Rajan is an associate professor at Fred Hutchinson Cancer Center in Seattle, Washington. She first started working with fruit flies during research for her master’s thesis. She became passionate about the organism’s scientific utility thanks to her mentor, and she continued working with Drosophila as she earned her Ph.D. in genetics from Baylor College of Medicine in Houston, Texas, and later as a postdoctoral fellow in genetics at Harvard Medical School in Boston, Massachusetts. Dr. Rajan has an NIGMS Maximizing Investigators’ Research Award grant supporting her research program focused on using fruit flies to investigate how adipocytes control brain function.
References
Brent AE, Rajan A. Insulin and Leptin/Upd2 Exert Opposing Influences on Synapse Number in Fat-Sensing Neurons. Cell Metab. (2020) doi: 10.1016/j.cmet.2020.08.017.
Kelly, KP, Alassaf M, Sullivan CE, Brent AE, Goldberg ZH, Poling ME, Dubrulle J, Rajan A. Fat body phospholipid state dictates hunger-driven feeding behavior. eLife. (2022) doi: 10.7554/eLife.80282.
